 Workstations / Ophthalmic tables
Workstations / Ophthalmic tables  Topography
Topography  Tomography
Tomography  Slit Lamp / Documentation
Slit Lamp / Documentation  Refraction Equipment
Refraction Equipment  Myopia Management
Myopia Management  Perimetry
Perimetry  Tonometer
Tonometer  Visual Test Equipment
Visual Test Equipment  Vitreoretinal Surgery
Vitreoretinal Surgery  Refraction
Refraction  Vision Test Types
Vision Test Types  Stereo Tests
Stereo Tests  Eye Test Units
Eye Test Units  Orthoptics / Pleoptics
Orthoptics / Pleoptics  Other Products
Other Products  Perimetry
Perimetry  Dry Eye
Dry Eye  Corneal Biomechanics
Corneal Biomechanics 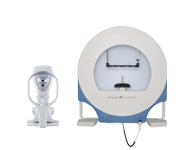 Glaucoma
Glaucoma 


The OCULUS Smartfield is a compact visual field device purposefully optimized for monitoring functional impairment in glaucoma. Based on an ultra-high-luminance LCD screen it performs standard automated perimetry of the central visual field and beyond. Despite its small size, the Smartfield perimeter offers a comprehensive clinical solution in visual field testing for any glaucoma practice.




| Programs | Pre-defined glaucoma, macula, screening and neurological tests user-defined tests |
| Test patterns | 30x24 (SPARK), 24-2, 10-2, customized patterns |
| Strategies | Threshold strategies: SPARK Precision, SPARK Quick, OCULUS Fast Threshold, Full Threshold (4/2), Age-adapted suprathreshold screening (2-zone, 3-zone) |
| Examination speed | Adaptive/fast/normal/slow/user-defined |
| Fixation control | Through central threshold, Heijl-Krakau (using the blind spot), live video image |
| Result display | Greyscale, dB values (absolute/relative), symbols, probabilities, 3D plot |
| Reports | Enhanced Glaucoma Staging System (GSS 2), Glaucoma Staging Program (GSP), PATH function-structure analysis, Threshold Noiseless Trend (TNT) progression report |


| Stimulus viewing distance | Infinity |
| Max. eccentricity horizontal/vertical | 30°/25° (60°/50° with fixation shift) |
| Stimulus size | Goldmann III |
| Stimulus colour | White |
| Stimulus duration | 200 ms/user-defined |
| Threshold range/step | 0.8 – 3 180 cd/m2 (2.5 – 10 000 asb), 0 - 36 dB/1 dB |
| Background luminance | 10 cd/m2 (31.4 asb) |
| Patient positioning | Height-adjustable measuring head, adjustable chin rest, double head rest |
| Software | Device control, patient management, backup and print software (Windows®) Built-in networking, easy EMR-integration, DICOM compatibility |


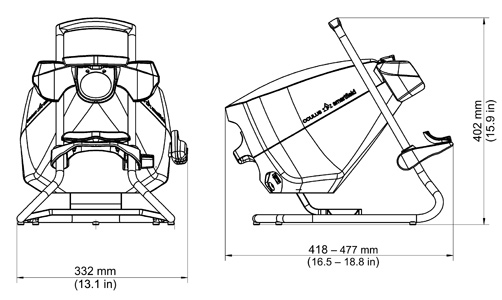
| Dimensions (W x D x H) | 332 x 418 to 477 x 402 mm (13.1 x 16.5 to 18.8 x 15.9 in) |
| Weight | 7.6 kg (16.8 lbs) |
| Max. power consumption | 30 W |
| Voltage | 100 - 240 V AC |
| Frequency | 50 - 60 Hz |
| Recommended computer specifications | Intel® CoreTM i5, 500 GB HDD, 8 GB RAM, Intel® HD Graphics, Windows® 10 |




In acc. with the Medical Device Directive 93/42/EEC

The OCULUS QM system is certified in accordance with ISO 13485 and (EU) 2017/745 (MDR).

Product Categories